- The following is a list of terms (and their definitions) commonly used in bioengineering:
- Deformation: Change in shape.
- Elastic deformation: A change in shape under load, which is completely reversed once the load is removed – intermolecular bonds are stretched (to allow shape change) but not broken.1
- Plastic deformation: A change in shape under load, which persists after the load is removed (i.e. permanent) – intermolecular bonds are broken and reformed elsewhere.1 Therefore in plastic deformation the object is given a new shape.
- Elasticity: The ability of a material/structure to return to its original shape, following deformation through loading.
- Tensile load: Collinear forces applied to a material/structure in order to pull it apart – causes lengthening of the object in the direction parallel to the force and shortening in the direction perpendicular to the force (see Figure 1a).
- Compressive load: Collinear forces applied to a material/structure in order to push it together – causes shortening of the object in the direction parallel to the force and lengthening in the direction perpendicular to the force (see Figure 1b).
- Direct force: Collinear forces applied along the axis of the material/structure – causes linear deformation. Tensile and compressive loads are both direct forces.
- Shear force: Unaligned forces applied away from the material/structural axis – causes angular deformation (see Figure 1c).
.png)
.png)
.png)
Figure 1. These diagrams demonstrate the effects of (a) tension, (b) compression, and (c) shear on an object.
- Stress: Force (F) applied to a material per unit area (A).
Units = N/m2
Normal stress (σ) – due to direct (tensile/compressive) forces.
Shear stress (τ) – due to shear forces.
Volumetric stress – normal and shear stress will always change the object’s shape. In volumetric stress equal compressive/tensile/shear forces are applied to an object causing its volume to change but its shape remains unaltered (e.g. a submarine’s volume will decrease in deep water due to hydrostatic compressive forces acting on all sides, but as all the forces acting on it are equal, the shape remains constant).2
- Strain: Quantative measure of material deformation. Direct strain (ε) = change in length (?L)/original length (L).
Direct strain has no units as it equates to a length (in m), divided by another length (also in m).
Shear strain (U) = angle of deformation (q). This can be difficult to measure directly as these angles are often very small. Therefore they can be calculated using the equation in Figure 2.
θ Angle is often measured in radians (Rad), not degrees. However, like direct strain, shear strain has no units as officially it represents the change in angle over original angle.
Volumetric strain – change in volume/original volume.2
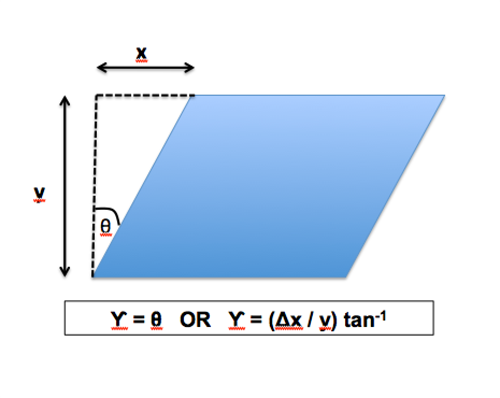
Figure 2. Figure showing how shear strain can be calculated.
- Elastic modulus (aka. Young’s modulus)
- Young’s modulus = material stiffness.
Young’s modulus (E) = stress (σ)/strain (∈).
This is a material property.
- Hooke’s law: The stress applied to a material is directly proportional to the resultant strain. Young’s modulus acts as the constant in the equation. Hooke’s law can therefore be written as:
σ = E∈
It is equal to the gradient of the stress/strain curve within the elastic zone (see Figure 3).1
Units = N/m2
NB. Some materials (e.g. polymers, biological tissue) demonstrate non-linear elasticity. This means the stress applied does not cause a proportional change in strain (i.e. it does not obey Hooke’s law); however, when the load is released the material returns to its original shape. These materials therefore do not have a measurable Young’s modulus. Instead a tangent modulus is taken from a chosen point on the stress-strain curve (see Figure 3b).1
.png)
Figure 3a. Stress-strain curve of material obeying Hooke’s Law, demonstrating how to calculate the Young’s modulus.
3b. Stress-strain curve of a material with non-linear elastic properties, demonstrating how to calculate the tangent modulus.
- Stiffness: Ability of a material/structure to resist deformation.
The stiffness of a material = ability to resist strain = Young’s modulus.
Stiffness of a structure = gradient of a load-displacement curve.
Units = N/m2.
- Strength: Ability of a material/structure to resist loading.
The strength of a material = ability to resist stress.
- Yield strength (aka yield point) – Stress at which a material permanantly deforms (usually referred to as the stress at which 0.2% plastic strain has occurred).
- Ultimate tensile stress – The greatest stress a material can withstand prior to failure. This is the highest point of a stress-strain curve.
Units = N/m2.
- Hardness: Resistance to point loading.
Point loading = load applied to a small localised area on a structure/material (e.g. the tip of a screw scratching the surface of a metal plate), as opposed to a load affecting the entire structure/material (e.g. from bending moments produced across a femoral plate when the patient bears weight).
In orthopaedics hardness usually refers to resistance to stratches or indentations on a materials surface.
- Toughness: Amount of energy a material can absorb prior to fracture.
Toughness = area under stress–strain curve.
Units = Joules/m3
- Ductility: The amount of plastic deformation permitted prior to fracture.
This includes the deformation seen in the plastic, strain hardening and necking regions (of a stress–strain curve) combined.
- Brittleness: opposite of ductility.
A brittle material allows little/no plastic deformation.
- Isotropic: The material/structural properties are dependant of the direction of loading.
E.g. Cortical bone = isotropic, as resists greater loads which are directed along the axes of the haversian systems.
- Anisotropic: The material/structural properties are independent of the direction of loading.
E.g. Woven bone = anisotropic, as the randomly arranged trabeculae allow near equal resistance to loading regardless of direction.
- Crystalline: Atoms arranged in a regular lattice formation.
E.g. Metals, some ceramics.
- Amorphous: Literally “without form” – there is no regular/organised arrangement of atoms.
E.g. Polymethylmetacrylate (PMMA/bone cement).