- The gold standard test in clinical practice is measurement of bone mineral density (g/cm3), of the vertebral spine and the hip. This is as recommended by the National Osteoporosis Society. Only vertebral measurements can be used to assess effectiveness of treatment at present.
When to measure bone mineral density:
- When there are risk factors present
- When it will alter management of a particular patient
Bone mineral density (BMD) measurement techniques:
DEXA scans
- Dual X-ray absorptiometry. Introduced in 1987.
- Simultaneous measurement of the passage through the body of X-rays with two different energies.
- Low radiation, accurate.
- Can be performed on vertebra, hip, whole body, or more peripheral areas.
- The National Osteoporosis Society has guidelines on the use of wrist DEXA scans.
- Wrist scans are less predictive of hip and vertebral fractures than hip and vertebra scans.
- If T score is above –1, reassure patient.
- If T score between –1 and –2, request DEXA of hip/spine.
- If T score is less than –2, treat, if clinically indicated. If monitoring required, refer for DEXA of hip/spine.
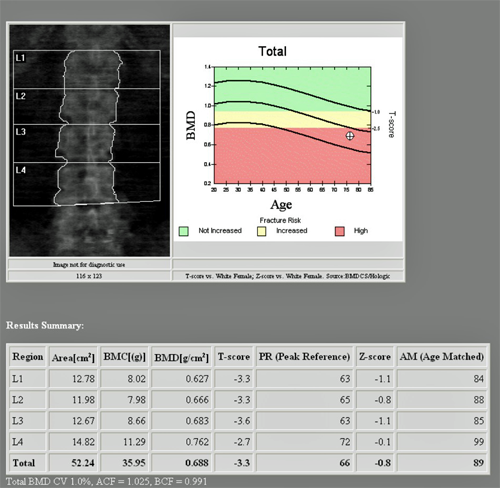
Figure 1. Dexa Scan Lumbar spine
Figure 2. Dexa Scan Proximal femur
Limitations of DEXA:
- DEXA scans only indicate a relative risk and cannot predict those who will actually experience a fracture.
- The presence of vertebral compression fractures or osteoarthritis or previous spinal surgery may produce a false negative result.
- Peripheral scans are useful to quantify fracture risk but only central scans can be used to monitor the effect of treatment.
Radiographic absorptiometry
- The first quantitative technique. Standard hand X-rays are taken next to an aluminium wedge. Bone mineral density is measured by comparison to the density of the aluminium. Only suitable for phalanges and metacarpals which are surrounded by only small amounts of soft tissue.
Single photon X-ray absorptiometry
- Still only useful in the peripheral skeleton, e.g. wrist as the body part needs to be placed within water, water bags or a water equivalent material to correct for overlying soft tissue.
Quantitative computer tomography
- An external bone mineral reference phantom is scanned along with the patient to calibrate the computed tomography measurements with bone equivalent values.
- More sensitive in vertebra than DEXA but less precision.
- Used for assessment of vertebral fracture risk, and follow up.
- However, much more expensive than DEXA and radiation dose higher.
Quantitative ultrasound
- Inexpensive, small, portable, does not involve radiation. Renewed interest in its use.
- The exact mechanism of the ultrasound interaction with bone is unclear.
- Different instruments will give different readings, due to differences in calibration, analysis software used.
- There is positive correlation with BMD of 0.7–0.9.
- Can be used to discriminate between normal and osteoporotic groups as effectively as DEXA.
However, the National Osteoporosis Society has published a statement saying that it supports the use of ultrasound in primary and secondary care and those found to have suspicious results should be considered for referral for DEXA scanning. It is not currently used to diagnose osteoporosis.
- Optional tests that may also be useful:
- Serum parathyroid hormone, 25- oral hypoglycaemic drug (OHD) levels, plasma electrophoresis.
- Bone marrow biopsy after tetracyline labelling, can rule out osteomalacia.
- Urinary free cortisol.
- Measurement of serum and urine markers of bone absorption and formation can be used:
Markers for bone formation: bone-specific alkaline phosphatase, osteocalcin.
Markers for bone resorption: collagen breakdown products in urine, e.g. N and C telopeptides.
T score
- The T score is the number of standard deviations (SD) above or below the mean peak bone mass for a sex and race-matched healthy normal young adultpopulation.
- This is the value the WHO takes its definition of osteoporosis from.
- The T score is approximately what the patient should have been at their peak bone density at about age 20 years.
Z score
- The Z score is the number of standard deviations below the mean for an age, sex and racematched population.
- As the Z score is measured against an age-matched group it cannot detect age-related osteoporosis.
- Elderly white women have weak bones even if the bone density is average.
- Low Z scores in young patients may suggest anorexia, chronic ill health or vitamin D deficiency.
Bone mineral density (g/cm2)
- This is most useful when values are compared to a previous earlier scan result after starting treatment to quantify any improvement.