Case based Discusson Limb Bud
Knowledge of the embryonic limb bud and its different zones and is essential to understand congenital limb deformities. The limb bud is made of mesoderm covered by ectoderm, and the upper limb bud appears toward the end of the third week followed by the lower limb bud which appears about 4 days later (fourth week).
The Apical Ectodermal ridge is the most distal zone of the bud (made of the covering ectoderm) and controls proximal to distal growth or elongation. Deep to the AER is the progress zone that is made of rapidly dividing mesoderm cells which enables this longitudinal growth. AER is controlled by the FGF gene family and injuries to AER will lead to congenital amputation, defects in the ridge can lead to truncation (such as club hand).
The AER appears first then the Zone of Polarizing Activity (ZPA) appears along the posterior aspect of the limb bud and signals the anteroposterior (radio-ulnar) growth. It is under the control of the SHH compound. Abnormalities in the ZPA can lead to a range of abnormalities from congenital absence of thumb (anterior / radial abnormalities) to ulnar polydactyly with SHH upregulation to loss of ulnar digits in the case of SHH downregulation (posterior /ulnar abnormalities).
The third signalling centre controls dorso-ventral growth comprises the rest of the ectoderm (the non AER area of ectoderm) and is controlled by the WNT gene.
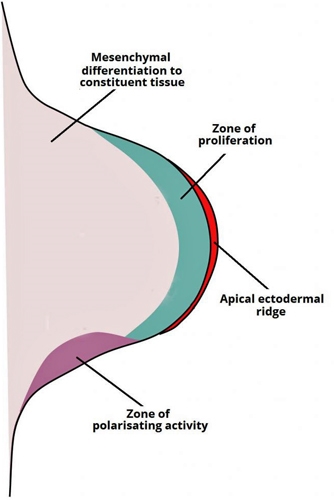
Figure 11.Limb elongation occurs through proliferation of the underlying mesenchyme core, in which the AER plays a crucial role in ensuring that the mesenchyme immediately underneath it remains undifferentiated. As growth proceeds, the proximal mesenchyme loses signals from the AER and begins to differentiate into the constituent tissues of the limbs.
Examiner: Please have a look at this X ray and describe what you can see.(Figure 11)
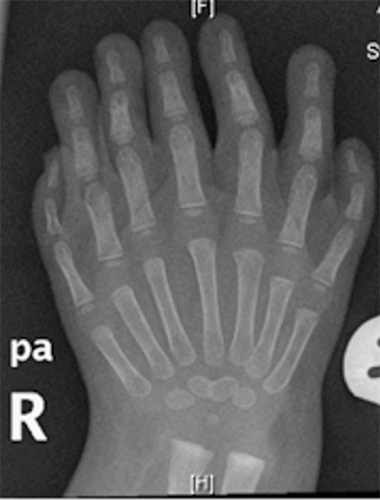
Figure 12.Radiograph hand
Candidate: There is symmetric polydactyly with absence of radial rays, these are usually features of ulnar dimelia or mirror hand which is a congenital condition.
Examiner: That is correct, what is the genetic defect that would lead to this
Candidate: Sonic hedgehog (SHH) gene, which controls the Zone of polarizing activity of the limb bud which in turn signals the Antero-posterior or radio-ulnar limb growth.
Examiner: If Upregulation of the SHH gene leads to ulnar duplication why do we see associated radial absence
Candidate: The posterior or ulnar components form earlier than the anterior or radial components so a disruption the antero-posterior signalling leading to an ulnar duplication due to upregulation of the SHH will also lead to losing the elements that form later ie the radial ones.