Non-steroidal anti-inflammatory drugs (NSAIDs)
- Historically, non-selective cyclooxygenase-1 (COX-1) and cyclooxygenase-2 (COX-2) inhibitors (often indomethacin) have been recommended for 6 weeks.
- More recently, a meta-analysis by Xue et al. (Int Orthop 2011) compared selective COX-2 inhibitors with non-selective COX-1 inhibitors in four randomised controlled trials. There was no difference in efficacy between the two.
- A Cochrane review assessed 16 randomised trials and found peri-operative NSAIDs were seen to reduce the incidence of HO by between one half and two thirds.
- Care needs to be taken with potential gastrointestinal side effects and delayed or non-union of an osteotomy has been performed.
Bisphosphonates
- Bisphosphonates block the aggregation, growth and mineralisation of calcium hydroxyapatite crystals.
- There is no conclusive proof of their efficacy in the prevention of HO.
Radiotherapy
- Prophylactic radiotherapy reduces HO after THA. A single dose of 700 cGy can be given either 4 hours preoperatively or within 72 hours (Figure 5). Both preoperative radiation and postoperative radiation were found to be equally effective at the hip, and no specific differences in complications were noted.
- The mechanism of action is thought to be due to inhibition of the fast-dividing osteoprogenitor cells that are present within the first week after surgery.
- These then differentiate into mature cell types such as osteoblasts. The lower mitotic rate of mature cells protects them from the damage to DNA caused by radiation.
- Radiation exposure is limited to the soft tissues immediately around the hip joint, and ingrowth surfaces must be appropriately shielded.
- Although no cases of malignancy after prophylatic radiation have been reported, this is a theoretical risk to consider.
- Additional side effects include progressive soft tissue contracture, delayed wound healing, non-union and inhibited ingrowth of press fit implants.
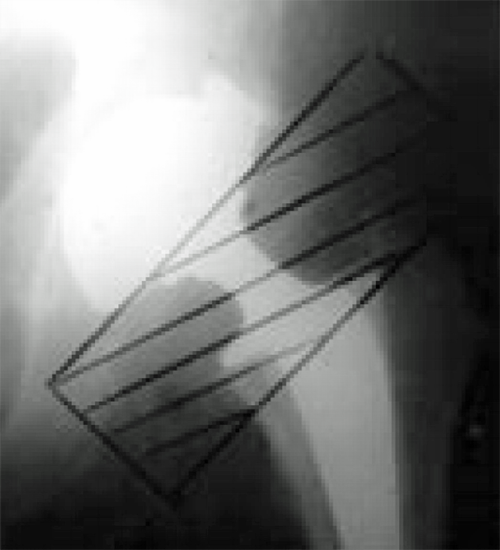
Figure 5. The radiation field after THA.
NSAIDs versus radiotherapy
- Pakos and Ioannidis (Int J Radiat Oncol Biol Phys 2004; 60: 888–895) performed a meta-analysis and suggested radiotherapy was more efficient than NSAIDs at preventing Brooker grade 3–4 HO.
- The overall difference in absolute risk was, however, small.
- Other studies have reported no difference between NSAIDs and radiotherapy in preventing heterotopic ossification.
- The decision to use radiotherapy or NSAIDs medication for prophylaxis can be tailored to individual patient conditions.
Combination therapy
- Radiotherapy and NSAIDs in combination may offer a potential advantage and three retrospective reviews have suggested lower incidence compared to single therapy, but further evidence is required.